Understand the Basics of Informed Consent
Informed consent is a vital ethical and legal principle that underlies all medical procedures, including stem cell treatments. It involves providing a patient with adequate information about a procedure, including its potential benefits and risks, alternative treatments, and the potential consequences of not undergoing the treatment. This empowers the patient to make an informed decision about their healthcare.
The National Library of Medicine regards informed consent as a cornerstone of patient rights, reinforcing the autonomy of the patient and ensuring that they are not subjected to any medical procedure without their voluntary approval.
Failure to obtain informed consent can lead to ethical and legal implications, such as accusations of misconduct, malpractice lawsuits, and potential damage to the doctor-patient relationship. Therefore, understanding this principle is imperative for both healthcare providers and patients.
The Concept of Stem Cell Treatment
Stem cell treatment, often heralded as the future of regenerative medicine, harnesses the body’s own repair kit to heal diseases and injuries. This innovative treatment uses the unique ability of stem cells to self-renew and differentiate into any cell type.
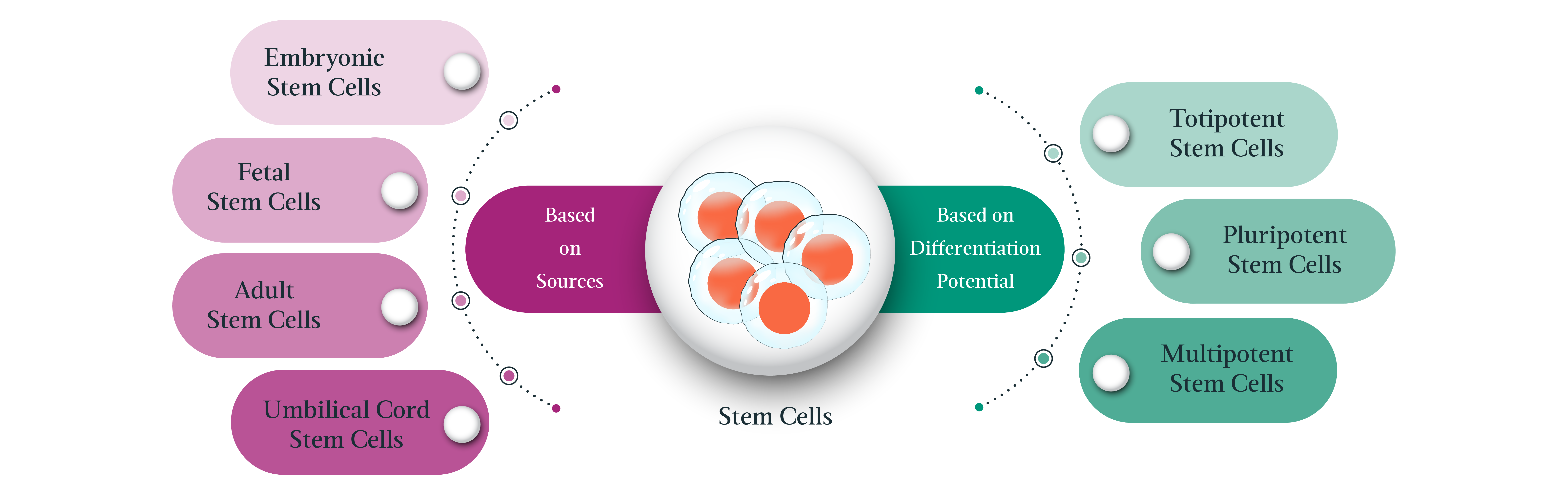
Types of Stem Cells and Their Sources
There are mainly three types of stem cells: embryonic stem cells, adult stem cells, and induced pluripotent stem cells. Embryonic stem cells are derived from embryos, adult stem cells are found in adult tissues, and induced pluripotent stem cells are adult cells genetically reprogrammed to an embryonic stem cell-like state.
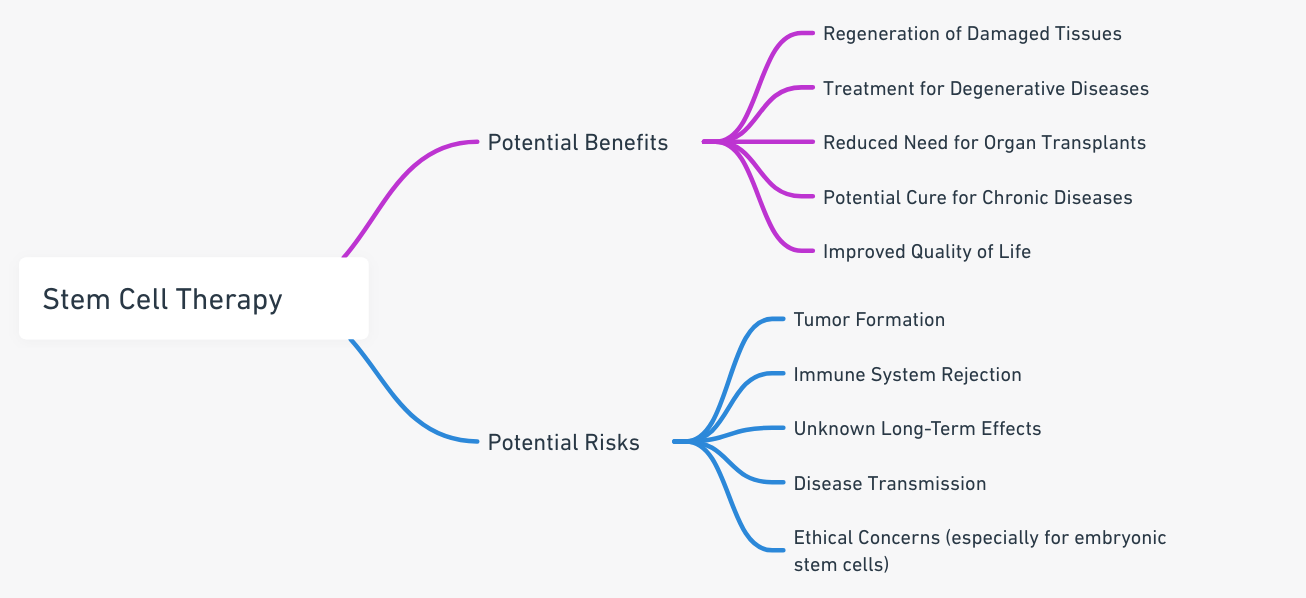
Potential Benefits and Risks of Stem Cell Treatment
Stem cell treatment holds immense potential for treating diseases like Parkinson’s, heart disease, and diabetes. However, it’s not without risks. Potential complications include reactions to the treatments, failure of cells to work as expected, and the possible development of cancer.
It’s pivotal to weigh these potential benefits and risks with your healthcare provider before pursuing stem cell treatment.
Understanding Informed Consent in Stem Cell Treatment
Informed consent is a crucial aspect of any medical procedure, including stem cell treatment. It protects both the patient and the healthcare providers by ensuring that the patient understands the potential risks and benefits of the treatment.
Potential Risks and Benefits
Like any medical procedure, stem cell treatments come with both potential benefits and risks. The benefits may include the potential to treat or even cure certain diseases. However, the risks can range from minor side effects to serious complications, such as rejection of the transplanted cells. It is imperative for these to be discussed thoroughly before the procedure.
The Physician’s Role in Ensuring Informed Consent
Physicians play a key role in the informed consent process. They are obligated to provide clear, comprehensive information about the treatment, enabling the patient to make a well-informed decision. This includes explaining the procedure, its potential outcomes, alternative treatments, and the potential risks and benefits associated with each. This information must be presented in a way that the patient can easily understand. The American Medical Association provides guidelines and resources to help physicians fulfill this critical responsibility.
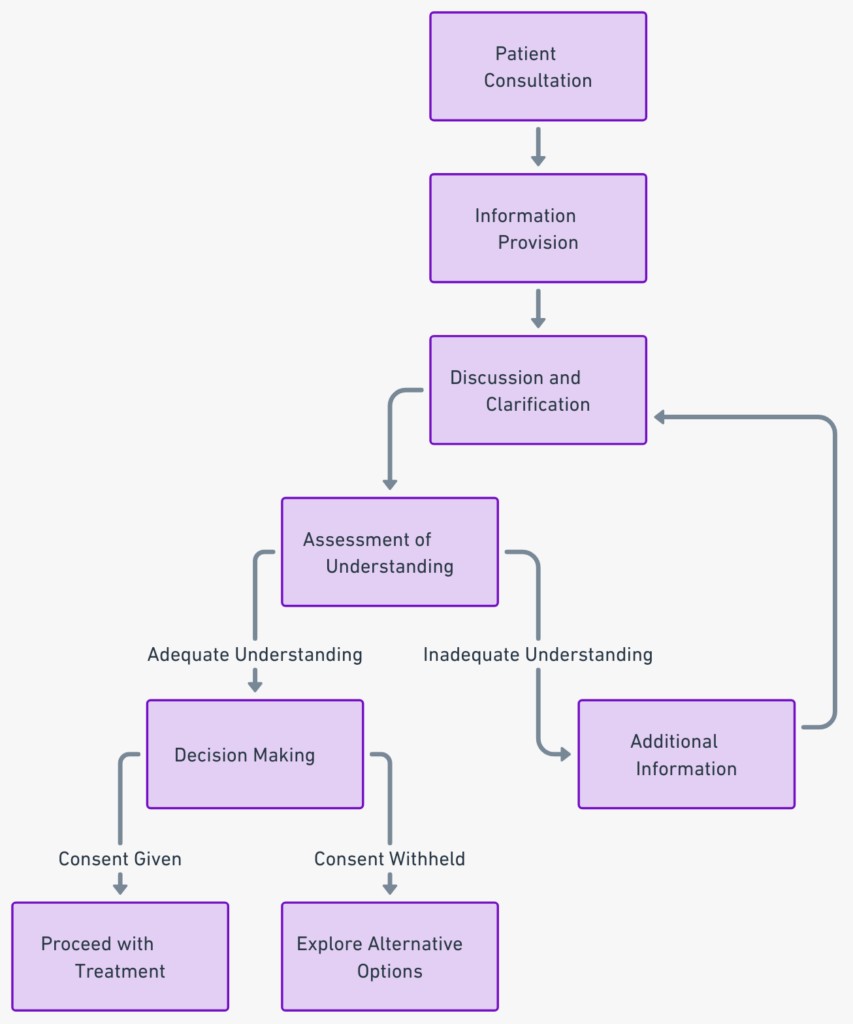
Key Components of Informed Consent in Stem Cell Treatment
Undeniably, informed consent is a significant part of medical procedures, especially in advanced treatments like stem cell therapy. It comprises four crucial components.
- Information about the Procedure: A comprehensive explanation of the stem cell treatment is the first step. This includes the purpose of the procedure, potential benefits, risks, and alternatives. This source offers a detailed insight into the process.
- Comprehension of Information: Ensuring the patient fully understands the information provided is essential. This entails clarifying any complex medical jargon and confirming the patient’s understanding.
- Voluntariness in Decision-Making: The patient must voluntarily agree to the treatment without any coercion. The decision should be the result of careful consideration of the provided information.
- Documentation of Consent: Finally, the entire consent process should be meticulously documented. This documentation serves as a legal record and a reference point for future discussions.
Understanding these elements is vital before undergoing stem cell treatment.
I recommend adding a flowchart to visually represent the informed consent process. This can enhance understanding and retention of the information.
Understanding Risks Associated With Stem Cell Treatment
The innovation of stem cell treatment brings about a new wave of medical possibilities. Yet, it is crucial to acknowledge the potential risks and side effects that come with this advancement. Common immediate side effects often include pain at the injection site, fever, and allergic reactions.
While these may be temporary, there are potential long-term effects as well. These can range from failure of the cells to work as expected, to the development of tumors [source].
Communicating Risks and Informed Consent
Informed consent is a cornerstone of patient rights, ensuring that individuals are aware of the potential risks and benefits of any treatment. In the context of stem cell treatment, clear communication about these potential risks is essential for patients to make an informed decision [source].
Ethical Considerations in Stem Cell Research and Therapy
Stem cell research and treatment offer potential solutions for a myriad of health conditions. However, they also raise significant ethical concerns that can cloud the informed consent process.
Controversies Surrounding Certain Types of Stem Cells
The primary controversy lies in the use of embryonic stem cells, as the extraction process often involves the destruction of the embryo, raising questions about the sanctity of life. This ethical dilemma has resulted in varying regulations across different jurisdictions.
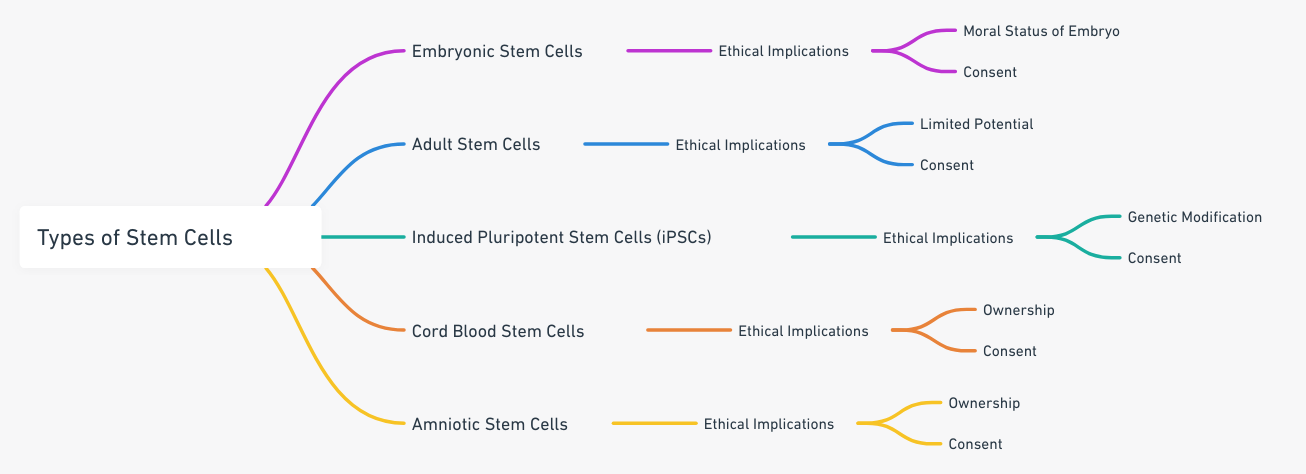
Ethical Implications of Stem Cell Research and Therapy
Other ethical implications include issues of justice and fairness. For instance, will everyone have access to such advanced therapies, or will they be reserved for those who can afford them? Informed consent also becomes complex when considering the potential unknown long-term effects of stem cell treatments.
The Informed Consent Process and Ethical Issues
Informed consent is a critical component of ethical medical practice. However, the inclusion of these ethical issues in the consent process can make it intricate. It is essential for patients to understand all the potential risks, benefits, and ethical implications before agreeing to treatment.
The Role of Regulatory Bodies in Stem Cell Treatment
Regulatory bodies play a crucial role in ensuring the safety, efficiency, and ethicality of stem cell treatments. Bodies like the FDA create and enforce regulations that govern the use and application of stem cells in medical settings. These regulations are designed to protect patients from potential harm, ensure that treatments are scientifically sound, and maintain public trust in the healthcare system.
Importance of Informed Consent in Stem Cell Treatment
These regulations not only ensure the safety and efficacy of stem cell treatments but also mandate that patients give informed consent before undergoing any procedure. Informed consent is a process that involves educating the patient about the benefits, risks, and alternatives of the treatment. This ensures that patients make knowledgeable decisions about their healthcare, respecting their autonomy and protecting them from potential exploitation.
Implications for Patients and Healthcare Providers
For patients, these regulations provide assurance of the safety and integrity of stem cell treatments. They also guarantee access to comprehensive information, enabling informed decision-making. Healthcare providers, on the other hand, are held to high standards of practice and ethics, ensuring they deliver the best care possible. This regulatory framework, thus, promotes trust, transparency, and accountability in the realm of stem cell treatment.
Empowering Patients: The Right to Know and Decide
In the realm of healthcare, patient autonomy is paramount. It’s the bedrock that empowers patients to make informed decisions about their treatment. It’s a crucial component of ethical medical practice. Especially in the context of stem cell treatment, where the stakes are high, informed consent is non-negotiable.
Securing Informed Consent: A Strategy for Patients
- Understand the Basics: Familiarize yourself with the nature of the treatment, its purpose, potential benefits, and risks.
- Ask Questions: Be proactive. A well-informed patient is an empowered patient.
- Be Assertive: Know that it’s your right to have all information before making a decision.
The Patient Rights Movement and Stem Cell Treatment
The ongoing patient rights movement continues to advocate for the right to informed consent, especially with emerging treatments like stem cell therapy. It ensures that the patients, not just the medical professionals, hold the steering wheel of their healthcare journey.
